Clinical Trials in Orbit: The Next Frontier of
May 06, 2026One Small Shift – Nutrition Without Restriction
May 05, 2026One Small Shift – Sleep Without Forcing It
Apr 08, 2026ONE SMALL SHIFT — Stress Without Fixing It
Mar 01, 2026Beyond the Benchmarks: Why 72% of Trials Fail
Feb 15, 2026What If Clinical Research Had Its Own Hippocratic
Feb 05, 2026Navigating Operational Realities in Clinical Trials Across Africa Beyond Strategy – Focus on Execution
- Abel Vissoukpo
- 4 Minutes Read Time
Africa is increasingly recognized as a key region for clinical research, offering diverse patient populations, high disease burden, and growing research capacity. However, while strategic interest is rising, operational realities on the ground are still often underestimated.
A gap persists between high-level trial design and field execution. Understanding and addressing these operational hurdles is critical to ensuring both trial success and data integrity.
What Are Some Common Operational Hurdles Sponsors Overlook When Developing Clinical Trial Strategies for Africa?
1. Site Variability Is Underestimated
One of the most common misconceptions is treating sites within a country or across the continent as operationally uniform. In reality, site capabilities vary significantly in terms of infrastructure, staff experience, patient flow, and data management practices.
Without tailored site assessments and support plans, this variability can lead to inconsistent performance and data quality issues.
Example:
In a multi-site study, an urban hospital may have electronic data capture systems and experienced coordinators, while a semi-urban site relies on paper-based records and limited staff. Applying the same monitoring intensity and timelines to both can lead to data inconsistencies and delays.
2. Centralized Decision-Making Slows Execution
Many sponsors rely on highly centralized processes for approvals and issue resolution. While this ensures control, it often does not align with the speed required in dynamic site environments.
Delays in responding to queries, protocol clarifications, or operational issues can quickly impact recruitment, compliance, and timelines.
Example:
A protocol clarification requested by a site may take several days to be validated at sponsor level. Meanwhile, patient visits are delayed, or deviations occur because site teams must act without timely guidance.
3. Training Is Not Sustained Over Time
Initial site training is typically well executed during study start-up. However, ongoing training, reinforcement, and practical guidance are often limited.
Given staff turnover and the complexity of protocols, continuous capacity building is essential to maintain compliance with ICH-GCP and ensure consistent trial conduct.
Example:
A trained study coordinator leaves mid-trial. The replacement receives minimal onboarding, leading to errors in informed consent documentation or data entry, which later trigger queries and potential audit findings.
4. Logistics and Supply Chain Constraints Are Underplanned
Transport of investigational products, laboratory samples, and essential materials can be affected by infrastructure limitations, customs processes, and geographic challenges.
When these factors are not fully integrated into planning, they can result in delays, protocol deviations, or compromised sample integrity.
Example:
Delays in customs clearance for investigational products or laboratory kits can interrupt patient dosing schedules or sample analysis timelines, directly affecting protocol compliance.
5. Patient Follow-Up Challenges Are Overlooked
Retention strategies are sometimes designed without fully considering local realities such as travel distance, financial constraints, and access to healthcare facilities.
This can lead to missed visits, increased loss to follow-up, and data gaps that affect study outcomes.
Example:
Patients living in rural areas may need to travel long distances for follow-up visits. Without transport support or flexible scheduling, missed visits and loss to follow-up increase, impacting data completeness.
6. Community Engagement Is Not Fully Leveraged
Trust plays a central role in patient recruitment and retention. Yet, strategies often focus more on digital outreach or site-level processes than on community engagement.
Sites that build strong local relationships consistently demonstrate better recruitment performance and patient adherence.
Example:
Sites that collaborate with local leaders or community health workers often see higher enrollment rates compared to those relying solely on hospital-based recruitment or digital campaigns.
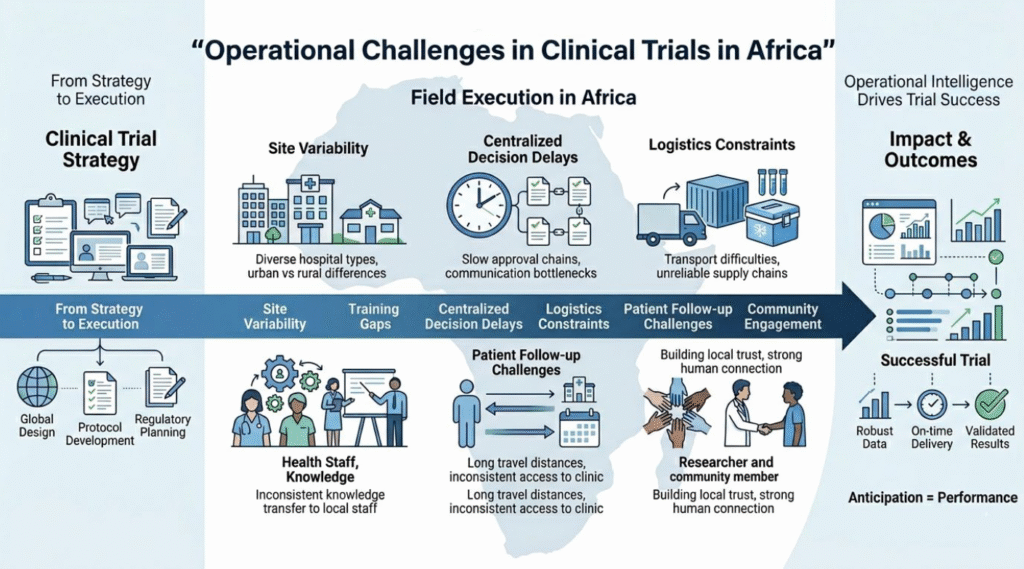
From Strategy to Execution: Bridging the Gap
Successful clinical trials across Africa require more than protocol compliance they require operational intelligence.
As emphasized in the literature, “context-adapted implementation is key to ensuring both efficiency and data reliability in global clinical trials” (NIH, Global Health Research perspective).
Sponsors must move beyond standardized approaches and invest in strategies that reflect field realities:
• Tailored site support
• Decentralized decision-making where appropriate
• Continuous training models
• Early integration of logistics planning
• Strong community engagement frameworks
Ultimately, clinical trial success in Africa is not determined at the design stage, but in execution. Anticipating operational hurdles is not a limitation it is a strategic advantage.
For more insights on clinical research operations, global trial strategy, and healthcare innovation, read more articles by Abel Vissoukpo at Interim Visits Author Page.
Abel Vissoukpo is a Clinical Research Associate (CRA) and Public Health Researcher passionate about transforming clinical research and public health initiatives into GCP-compliant, patient-centered, and data-driven outcomes. With solid experience in Clinical Research Operations, Healthcare Project Coordination, and Clinical Monitoring, he is committed to delivering high-quality research, upholding ethical standards, and driving measurable impact.
Over the years, Abel has coordinated and contributed to health and clinical research projects benefiting more than 500 patients. He has also implemented digital and AI-driven solutions that improved project efficiency by 30%, strengthening operational performance and overall research outcomes.















